Which Combination of Elements Represents Part of a Group, or Family, of the Periodic Table
The Periodic Chart of Table of the Elements
Overview
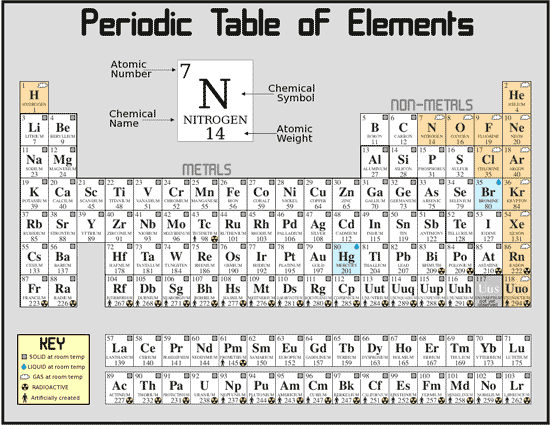
The Periodic Chart of the Elements is simply a way to arrange the elements to testify a big amount of information and organization. Every bit you read beyond the nautical chart from right to left, a line of elements is a Period. Equally you lot read down the nautical chart from top to bottom, a line of elements is a Group or Family. We number the elements, outset with hydrogen, number one, in integers upwards to the largest number. The integer number in the box with the chemical element symbol is the atomic number of the element and also the number of protons in each cantlet of the element.
History of the Periodic Tabular array
The periodic table is a way to organize the elements based on their similarities. The showtime version was constructed past Dmitri Mendeleev in 1869. He put the elements into a grid with increasing atomic weights and noticed the elements in each column had like chemic properties. Mendeleev was able to predict the existence of elements before they were discovered or synthesized past looking at gaps in his table. However, he too found inconsistencies. For example, according to Mendeleev'due south table, Argon should have been placed earlier Potassium. The solution to this problem came in 1913 when Henry Moseley organized the elements by diminutive number (the number of protons an atom has). He did this by shooting electrons at atoms and analyzing emission x rays. This new organization stock-still all of the contradictions created past the the atomic weight organization. Below is a picture of the final result.
Structure of the Periodic Table
The periodic table is structured in a manner that is very useful for chemists. The horizontal rows are called periods.
Typically the element's symbol, atomic number, and atomic weight are shown. An element's period tells united states of america the highest
unexcited energy level for an electron. Nosotros tin can tell the number of valence (outer) electrons besides as other useful
properties an element has from its group number. This volition be discussed more in the adjacent section. The periodic table
is too organized into four blocks according to the subshell in which the last electron resides. The s cake contains
the get-go two groups, the p block contains the last half-dozen groups, the d cake contains groups 3 to 12, and the f block
has the lanthanides and actinides ordinarily located in 2 rows at the bottom of the menstruation table. Blocks help when writing
out the electron configuration of an element.
Groups
In that location are ten named groups in the periodic table. All elements in a group have like properties. It is also possible
to tell how many valence electrons elements in certain groups take. The common group names and valance electrons are
outlined below:
| Group Number | Group Name | Valence Electrons |
| 1 | Alkali Metals | 1 |
| 2 | Alkaline metal World Metals | 2 |
| 3-12 | Transition Metals | varies |
| 13 | 3 | |
| xiv | 4 | |
| 15 | v | |
| 16 | vi | |
| 17 | Halogens | 7 |
| 18 | Noble Gases | viii |
A few points should exist noted. Although helium is in group 18, it only has 2 valence electrons. Likewise, hydrogen
is a nonmetal. In social club to predict the number of valence electrons that transition metals take, other methods
must exist used.
Trends of the Periodic Tabular array
There are also specific trends that the periodic tabular array follows. These deal with the size, ionization free energy,
electronegativity, and electron affinity of an element. The size (atomic radius) of an electron decreases
from left to right. This is because each time a proton is added the electrons are drawn closer in towards the
center. The atomic radius increases from peak to bottom. The ionization energy of an chemical element, or the amount of energy
required to remove an electron increases from left to correct and decreases from top to lesser. Electronegativity, the
tendency of an element to attract electrons, follows the same pattern as ionization energy. Electron affinity also shows
trends, only they are less consistent than the others. Electron affinity is the amount of energy released when an electron
is added to a neutral atom to form a negative ion. Information technology generally increases left to right and decreases pinnacle to bottom.
One concluding major trend of the flow table deals with the metal character of an chemical element. Elements with lower ionization
energies, electron affinities, and electronegativity have stronger metallic characteristics. This ways it decreases left
to right and increases top to lesser which helps explain the staircase shape in many periodic tables that metalloids create.
Future Changes to the Periodic Tabular array
The periodic table is not set in stone. New elements will be discovered, and scientists are not sure whether or not the
periodic table will continue with the current trends. Adjustments may demand to exist made. Also, the number of possible elements
is debated. Current predictions range from 126 to 155 elements.
PROPERTIES OF Thing
Physical Properties of Matter
Thing is anything that has mass and takes upwardly infinite. Information technology includes all the physical "stuff" around us—rocks, houses, water, air, people, ants, trees, etc. All matter can exist every bit a solid, a liquid, or a gas. Affair includes all the things we tin can weigh (to find the mass) and measure out to find the volume (considering they take upwardly infinite).
Different substances—dissimilar types of thing—take different properties that can be used to distinguish them from each other. These properties are things that y'all can observe or measure near the substance. For instance, water and isopropyl alcohol (rubbing alcohol) are both clear, colorless liquids at room temperature. However, they can be told apart because they take other properties that are different. Water is odorless while the isopropyl alcohol has a stiff smell. The booze evaporates very speedily while the water evaporates slowly. Too, the alcohol is flammable—information technology can be attack burn with a match (but don't endeavour this at home!). Water, on the other hand, is not but inflammable; it is used to put out fires.
The backdrop of matter can be divided into ii categories: physical properties and chemical properties. Physical backdrop can be observed or measured without changing the composition of the substance. An example is odor. When you odor h2o or isopropyl alcohol, neither of these substances changes into something else. They remain water and isopropyl booze. Similarly, when water and isopropyl booze evaporate, they practice not change into something else. They move from liquid into gas, but the water is still water and the booze is all the same alcohol. Considering these substances practise not modify into different substances, the properties of scent and speed of evaporation are physical properties.
Hither are some other examples of concrete properties. Copper is a substance that is a solid metal at room temperature with a melting point of 1083°C. It is shiny, bendable, and orange-brown-colored. Information technology tin can be flattened into a very sparse sail of metal or information technology can be stretched into a sparse wire. It can conduct estrus quickly and information technology can conduct electricity, which is why copper is used in power cords and other wires.
All of these properties of copper are concrete backdrop because the copper does not change into anything else when these properties are observed. You tin can see that information technology is a solid and shiny without doing anything to it. You can curve, flatten, or stretch, it. These actions will change the shape and appearance, simply it remains copper. Even heating it until it melts into a liquid or running electricity through it volition not change the copper into some other substance.
As mentioned above, some backdrop are chemical instead of concrete properties. In this blazon, the substance actually changes into another kind of substance. The flammability of isopropyl alcohol is an example of a chemical holding. When alcohol is burned, the alcohol actually turns into something else. At the end of the change, the alcohol is gone and has been replaced by carbon dioxide (a colorless gas) and water vapor (water that is in the gas land). This is not a physical property considering the isopropyl alcohol is changed into something else.
Chemic Properties of Affair
Chemical backdrop are observable characteristics of the makeup of a substance. Observable does not necessarily mean that one can visually see information technology; rather it means that we can note the effect. We tin observe a aroma, a voltage, or even a time.
Chemical properties are observable characteristics of the makeup of a substance.
We find out that Silver (Ag) reacts with elemental Oxygen (O2) to grade Silverish Oxide Ag2O. If this necklace were actually plated with silver instead of being made out of silver nosotros would expect a different outcome than what we observe with pure silver. Ane chemical element that we have an abundance of in our atmosphere is Oxygen (O2), so we could do a lot of tests information technology. Nosotros leave the necklace in a canteen and fill it with pure O2. After some time, we see that the necklace has reacted. We accept out the necklace and analyze the chemical composition of the consequence; we detect that we take a practiced corporeality of Silver Oxide (Ag2O) nowadays. Smashing, chemic properties accept helped usa solve the mystery of the shiny necklace, but besides bad we no longer have our necklace. That's a main betoken that I should tell y'all, in the process of observing chemical properties we often must alter the chemical composition of the original substance to obtain the property. With this said, make sure not to get and react your mom'due south necklace without letting her know, because the reaction is often irreversible!
Now that we learned what chemical properties are, let's get back to nuts "The Periodic Table of Elements." Of course in that location are many ways that we tin can allocate these elements, we can allocate them according to size, color, smell, behavior, or even by the style they react with water.
Chemists have adamant that the best style to organize the elements is to align them into vertical columns ("groups" or "families") then to further allocate them into regions based on their behavior.
Permit's start off by exploring the families of the periodic table:
Grouping 1A: Brine Metals (Li, Na, K, Rb, Cs, Fr)
All of these elements are metals and exist as solids at room temperature. These elements are extremely reactive, due to their valence shell containing only 1 electron. They all react with water to produce hydrogen and alkaline solutions. In instance yous're wondering, alkaline solutions are mixtures of a basic solid dissolved in water. Being that these elements absolutely detest being alone, we always find these elements reacted with another element in nature. An example of this is sodium chloride (NaCl).
Grouping 2A: Element of group i Earth Metals (Be, Mg, Ca, Sr, Ba, Ra)
Merely like Group 1A metals, these elements (with the exception of Exist) react with h2o to produce element of group i solutions. Many times these elements are establish as their cousin oxides, such as Calcium Oxide (CaO), in which instance they even so react with h2o to produce alkaline solutions. Magnesium and Calcium are the globe'south seventh and fifth about abundant crust constituent. No wonder they are called "earth metals".
Group 3A: (B, Al, Ga, In, Tl)
The main similarity of these elements is that they all class compounds with analogous formulas. What does that hateful? Well if we take Boron (B) and mix it with affluence with Chloride (Cl), nosotros end upward with BCl3. Ok, I agree, not too interesting; but, what if we take another chemical element of group 3A…how virtually Aluminum (Al). If Aluminum is mixed with an affluence of Chloride nosotros get (drumroll please) AlCl3. This is pretty boggling, so because of this we tin can merits they are all a family and react similarly.
Group 4A: (C, Si, Ge, Sn, Pb)
Starting with this grouping and moving to the right, we outset to see more and more than nonmetals present in each family. In this family, Carbon (C) is a nonmetal, Silicon (Si) and Germanium (Ge) are metalloids, Tin (Sn) and atomic number 82 (Pb) are metals. It becomes credible that families are becoming more diverse moving to the correct. Since this family, 4A, happens to be very diverse, more variation occurs in the properties of this group. Only every bit in group 3A, all of these elements happen to form like compounds such every bit CO2, SiO2, GeO2, SnO2, and PbO2.
Group 5A: (N, P, As, Sb, Bi)
Again, nosotros meet a metal mixed with a nonmetal and a metalloid in this family unit…it appears that there probably won't exist that many backdrop that these elements share. It turns out that these elements are grouped together because they all course similar compounds. An instance is in the case of Oxides, N2O5, P2O5, and As2O5.
Grouping 6A: (O, South, Se, Te, Po)
Like our families at home, group 6A is a very diverse family unit of elements. These elements don't have a whole lot in common, just then again what do you really take in common with your parents?. All elements in grouping 6A form similar Oxygen containing compounds SO2, SeO2, and TeO2. Interestingly, they all form similar sodium containing compounds as well, Na2O, Na2S, Na2Se, and Na2Te.
Group 7A: Halogens (F, Cl, Br, I, At)
This entire grouping is formed of nonmetals indicating that we are likely to discover a lot of similarities within this family unit. All of the halogens exist as diatomic molecules in nature, are extremely reactive (the most reactive of all the elements, in fact), and they all react extremely tearing with the brine metals to form salts. Enjoy your table common salt (NaCl) because sodium reacted pretty violently with Chloride to form this.
Grouping 8A: Noble Gases (He, Ne, Ar, Kr, Xe, Rn)
The proficient onetime noble gases! Where would we be today without some helium balloons for your birthday or no neon lite up signs? All of these gases are extremely rare on globe and therefore were not discovered until late 19th century. These elements absolutely hate to react with other elements. They prefer to exist equally isolated every bit possible because, after all, they are noble.
Groups 1B-8B: Transition Metals
All of these elements are metals, hence the name transition metals. Information technology'southward difficult to discover similar properties in these groups so nosotros will exit them lone for at present. The master classification that all these elements share is their unfilled d-subshell. Nosotros didn't talk too much most quantum numbers, simply you will soon see how valence shells can touch on the behavior of elements and how they react.
Then allow's all give a circular of applause for Dmitri Ivanovitch Mendeleev and his organization of the modern mean solar day Periodic Table of Elements. Mendeleev saw that elements possess similar characteristics (properties) if looked at by a office of their atomic number. This became known as "The police of chemical periodicity" it'due south pretty obvious why. This law states that the properties of elements are functions of the atomic number of the chemical element, WOW! Even more interesting, a lot of the elements were not fifty-fifty discovered when he made the first periodic table of elements, so he actually left spots in his original table for "possible elements" that he believed would exist discovered in the time to come. Now that's cool!
Some more interesting chemical properties
It turns out that chemistry is filled with tons and tons of chemical properties. Some properties are shared between substances and some are not. It turns out that there are and so many different chemical compositions in our universe that it would be impossible to approach each separately. Luckily for us, chemists accept taken individual compounds and grouped them into different classes that each share like chemical backdrop.
Permit's talk about a few.
An alcohol is any organic compound that contains a hydroxyl grouping attached to a carbon. Every bit you can imagine, there is a huge number of ways that one tin can adhere a hydroxyl grouping to a carbon cantlet. There are so many ways that chemists realized, it's probably smarter to just classify them all as types of alcohols.
All alcohols share some common chemical properties, for example upon reacting an alcohol with O2 (too known as a combustion reaction) nosotros volition e'er become carbon dioxide (CO2) and water (H2O).
Another interesting holding is booze's ability to be oxidized. Alcohols can exist oxidized into carboxylic acids with the usage of oxidizing agents such as potassium dichromate (VI) or potassium manganite (VII). By the way, this is the reason wine turns sour if left in the presence of oxygen.
Nosotros can go on and on about the chemical backdrop of various classes, but the chief point of this commodity is to go you lot acquainted to what a chemical belongings is.
What'south the big idea?
So what? I know what a chemic property is, how does that aid me? Well, simply like identifying your friends by their respective names, just like being able to tell the deviation between solar day and night, and merely like beingness able to differentiate betwixt hot and common cold, as maturing chemists we need the ability to identify a substance and set it apart from every other substance in the universe! Nosotros tin can't go around claiming ethanol is the same every bit water simply considering they share the same property of colour. We can't breathe in carbon monoxide (CO) instead of Oxygen (O2) because they both are odorless. The more chemical properties we know virtually a certain type of affair, the better we can differentiate that substance from every other substance in our universe!
Backdrop of Affair Do Quiz
Which of these is
nota physical holding of h2o?
A.
boiling indicate of 100°C
B.
colorless liquid at room temperature
C.
separates into oxygen and hydrogen gases due to electricity
D.
is able to erode rocks over time
The correct reply hither would exist C.
The water is turning into oxygen and hydrogen; therefore, the chemical makeup is changing due to it breaking apart. After the procedure described, it is no longer water.
Which of these is a physical property of the metallic aluminum, out of which aluminum foil is fabricated?
A.
Aluminum foil can be crumpled into a ball.
B.
Aluminum tin can be corroded by oxygen to form aluminum oxide.
C.
Aluminum tin exist "destroyed" past hydrochloric acid (HCl), forming aluminum chloride and hydrogen gas.
The correct respond hither would be A.
The aluminum is only changing shape; in the other choices, the aluminum is irresolute into other substances.
True or fake? When water is heated to its boiling point of 100°C, it bubbles and appears to disappear into the air. This is an instance of a concrete property.
The correct answer here would be A.
The h2o is changing phases from liquid to gas, just it is still water
Which of the following is a chemical property?
A.
The summit of a person
B.
The colour of Mountain Dew
C.
A carboxylic acid is formed with the reaction of an alcohol with oxygen.
D.
None of the above
The right answer here would be C.
Why exercise we identify substances by their properties?
A.
We don't.
B.
To differentiate that substance from other substances.
C.
To help us acquire more about affair.
D.
Both B and C
The correct answer here would exist D.
How tin we tell the difference between h2o and ethanol?
A.
past the color.
B.
by feeling them.
C.
by reacting them with oxygen.
D.
None of the in a higher place.
The right reply hither would exist C.
What is the definition of chemical periodicity?
A.
Backdrop of elements are functions of mass number.
B.
Properties of elements are functions of catamenia.
C.
Properties of elements are role of atomic number.
D.
None of the to a higher place.
The correct answer here would be C.
Which of the following would react with h2o to form hydrogen and alkaline metal solution?
A.
Group 1A
B.
Group 4A
C.
Group 5A
D.
Transition Metals
The right answer here would be A.
Periodic Properties
The periodic chart came virtually from the idea that we could accommodate the elements, originally by diminutive weight, in a scheme that would show similarity among groups.
The original thought came from noticing how other elements combined with oxygen. Oxygen combines in some way with all the elements except the inert gases. Each cantlet of oxygen combines with two atoms of any element in Group 1, the elements in the row below lithium. Each atom of oxygen combines one-to-one with any element in Grouping 2, the elements in the row below beryllium. The group of transition elements (numbers 21-30 and 39-48 and 71-80 and 103 upwards) have never been adequately placed into the original scheme relating to oxygen. The transition elements vary in the ways they tin can attach to oxygen, but in a mode that is not and then readily apparent by the simple scheme. Grouping three is the group below boron. All of these elements combine with oxygen at the ratio of iii to two oxygens. Group 4, beginning with carbon, combines two to one with oxygen, etc. Gallium, element number 30-1, is the crowning glory of the Periodic Nautical chart as first proposed past Mendeleev.
Dmitri Ivanovich Mendeleev get-go proposed the idea that the elements could be arranged in a periodic way. He left a space for gallium below aluminum, naming it eka- aluminum, and predicting the properties of gallium fairly closely. The chemical element was found some years later just as Mendeleev had predicted. Mendeleev too accurately predicted the backdrop of other elements.
Most Periodic Charts have two rows of fourteen elements below the main body of the chart. These two rows, the Lanthanides and Actinides really should be in the chart from numbers 57 – 70 and from 89 – 102. To show this, there would have to be a gulf of fourteen chemical element spaces between numbers 20 – 21 and numbers 38 – 39. This would make the chart near twice equally long equally it is at present. The Lanthanides belong to Period 6, and the Actinides belong to Catamenia seven. In basic Chemistry courses you will rarely notice much use for whatsoever of the Lanthanides or Actinides, with the possible exception of Chemical element #92, Uranium. No chemical element greater than #92 is found in nature. They are all man-made elements, if you would like to call them that. None of the elements greater than #83 have whatsoever isotope that is completely stable. This means that all the elements larger than bismuth are naturally radioactive. The Lanthanide elements are so rare that you are not likely to run across them in well-nigh beginning chemistry classes. Another oddity of the Periodic Chart is that hydrogen does not really belong to Group I — or whatsoever other grouping. Despite existence over seventy percent of the atoms in the known universe, hydrogen is a unique element.
PERIODIC CHART OF THE ELEMENTS
Element, Ion, and Compound Symbols
Overview
Atoms, and their subatomic particles, are measured in a.grand.u., or atomic mass
units. An atomic mass unit of measurement is the equivalent of 1/12 of the mass of a carbon-12
atom. Elements are most often measured in a.m.u., grams, or moles. Each of these
measurements volition be further explained in the post-obit pages, every bit needed.
The Periodic Table
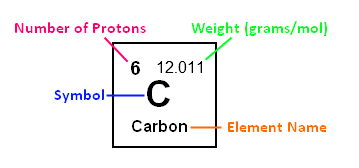
If you look up an element on the periodic table, you volition see the following:
Or, information technology might only be displayed this way:
The post-obit useful information is provided on about periodic tables:
Symbol: This is often a one or ii letter abbreviation for the element; for
example, carbon's symbol is C. When writing the symbol, the commencement letter of the alphabet is always
capitalized, and if information technology has a second letter, it is lower-example.
Element name: This is what the element is called. Some periodic tables have
both the symbol and the proper name, while others just take the symbol, so it is important
to know both.
Atomic Number: This is the number of protons the element contains. For instance,
carbon'south atomic number is 6, because there are half-dozen protons in a carbon atom.
Atomic Weight: This is often the decimal number given, and it is usually
in units of grams/mol. This number will be used profoundly in gram to mol conversions
later on. For example, carbon's diminutive weight is 12.011 thousand/mol.
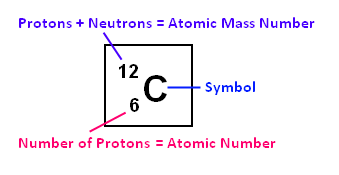
Diminutive Mass Number: This is a whole number that is usually very close to
the atomic weight. The atomic mass number is the number of protons plus the number
of neutrons in an atom. For instance, carbon'south diminutive mass number is 12, which means
it has 6 protons and 6 neutrons. This is helpful in calculating mass ratios.
For every element at that place is one and but 1 upper instance letter. There may or may non be a lower example letter of the alphabet with it. When written in chemical equations, we stand for the elements past the symbol lonely with no charge attached. The seven exceptions to that are the vii elements that are in gaseous form equally a diatomic molecule, that is, two atoms of the same element attached to each other. The list of these elements is best memorized. They are: hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine. The chemical symbols for these diatomic gases are: H2, N2, O2, F2, Cl2, Br2, and I2. Under some weather oxygen makes a triatomic molecule, ozone, O3. Ozone is non stable, and so the oxygen atoms rearrange themselves into the more than stable diatomic grade.
The diatomic gases (hydrogen, nitrogen, oxygen, fluorine, chlorine, bromine, and iodine), the Group one elements (lithium, sodium, potassium, rubidium, cesium, and francium), the Grouping two elements (beryllium, magnesium, calcium, strontium, barium, and radium), Group 7 elements, the halogens, (fluorine, chlorine, bromine, iodine, and astatine), and the noble gases (helium, neon, argon, krypton, xenon, and radon). If nil else, learning these as a litany will help you lot distinguish between radium, a Group 1 element, and radon, an inert gas.
Groups of two or more element symbols attached to each other without any accuse on them signal a compound. CaCl2 is a compound with ii chlorine atoms for each calcium atom. CuSO4 · 5H2O, cupric sulfate pentahydrate, is also a compound. It has 1 copper atom and one sulfate ion consisting of a sulfur atom and 4 oxygen atoms fastened to five molecules of water.
Charged particles, called ions, when written with symbols will have the charge, either positive (+) or negative (-), written to the correct and superscripted to the chemical symbol. For example, Na+ is the symbol for the sodium ion. Atoms or polyatomic ions with charges of more than than one, either positive or negative, accept a number with the charge. For instance (CO3)2- is the symbol for the carbonate ion. The carbonate ion has one carbon atom in information technology, three oxygen atoms, and a charge of negative two. Detect that the charge is outside the parentheses, indicating that the charge is from the polyatomic ion as a whole.
Ions and Isotopes
All naturally occurring atoms (of the elements listed on the periodic table) are
made upwards of neutral atoms, meaning they have the same number of protons and electrons.
An imbalance of protons and electrons creates an ion, an atom that contains
more than protons than electrons, or vice versa. Ions are created past the proceeds or loss
of electrons. Gaining an electron would give an ion a negative accuse, because electrons
make the atom more than negative. Losing an electron would give the ion a positive charge,
considering taking away the electron (which is negative) would make the cantlet more positive.
A positive ion is called a cation, and it can have a +ane, +ii, or +3 charge. A negative
ion is called an anion, and it can accept a -1, -2, or -3 charge. Sometimes the charges
may be college than positive or negative iii, but generally they are either + or –
1, 2, or 3.
Isotopes are as well important when determining mass and charge. Some elements
have isotopes, which ways that they have dissimilar types of atoms—some of which
have more neutrons than others. Now remember, neutrons practice not have a accuse, so
an isotope does non have a different accuse than that of the original elemental
atom, information technology simply has a unlike number of neutrons, which changes the atomic mass
slightly. For example, the most oftentimes occurring form of carbon is carbon-12, which
means that that form of carbon has half-dozen protons, 6 electrons, and half dozen neutrons. However,
some common isotopes of carbon are carbon-xiii and carbon-xiv, which can be interpreted
as follows: carbon-13 would take 6 protons and vi electrons (every bit usual) but this isotope
would have 7 neutrons, as xiii – six = 7. Carbon-14 would have 6 protons and 6 electrons,
as usual, but this isotope would have 8 neutrons, every bit xiv – six = 8.
Sometimes, compounds tin can be ions besides. Remember, nosotros said ions were elements with
a positive or negative charge. Well, ions can be made upwardly of compounds likewise. For
example, NO3 – is the negative nitrate ion that bonds with
hydrogen to class HNO3. Ions that are compounds are chosen polyatomic
ions, because they are made up of more than one atom of elemental material.
Categories of Elements
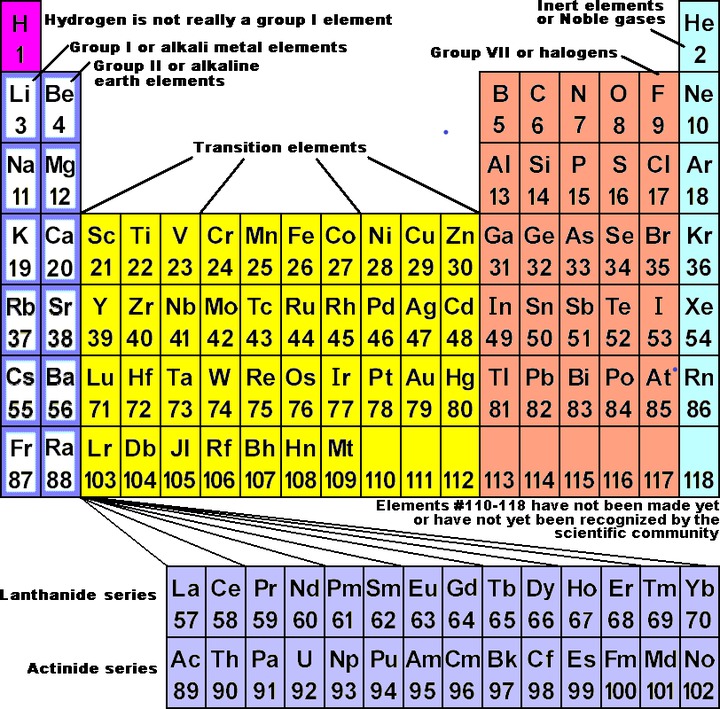
What Wyzant calls 'categories of elements' include; metals, non-metals, semi-metals, noble gases, and hydrogen.
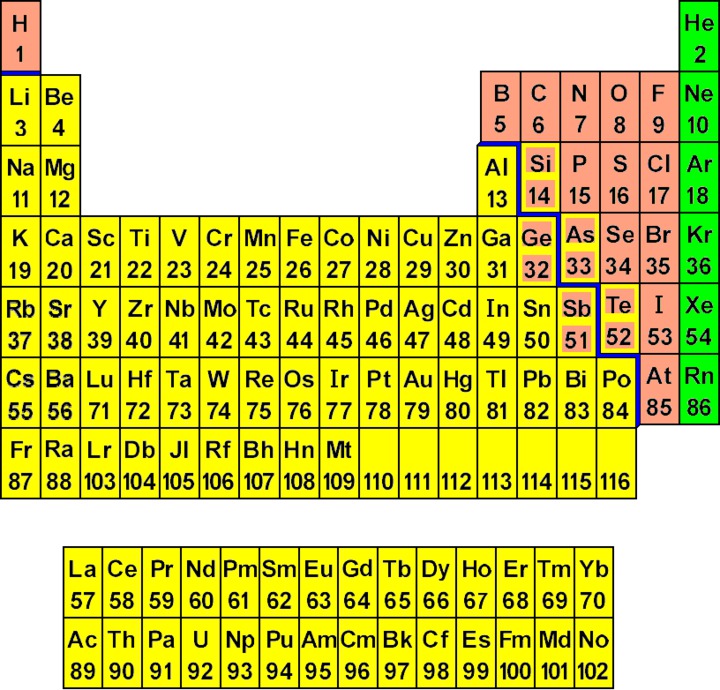
Consider a staircase-shaped line on the Periodic Chart starting between boron and aluminum turns to be between aluminum and silicon then down between silicon and germanium, between germanium and arsenic, betwixt arsenic and antimony, betwixt antimony and tellurium, between tellurium and polonium, and between polonium and astatine. This is the line between metal and non-metal elements. Metal elements are to the left and downwards from the line and non-metal elements are to the right and up from the line. Well, that's not exactly truthful. There is a line of non-metallic elements, Group viii, or Grouping eighteen, or Group 0, whichever way you count them, the noble or inert gases that are really an entire Grouping and category to themselves. Hydrogen is a unique element, the only member of its own Group and category.
Noble Gases
The noble gases, or inert gases, accept the following properties: For the nigh role, they exercise not make chemic combinations with any elements. There have been some compounds made with the noble gases, only only with difficulty. In that location are certainly no natural compounds with this grouping. They are all gases at room temperature. They all have very low boiling and melting points. They all put out a colour in the visible wavelengths when a low pressure of the gas is put into a tube and a loftier voltage current is run through the tube. This type of tube is called a neon calorie-free whether the tube has neon in it or not. The inert gases are non-metals because they are not metals, but they are significantly dissimilar from the other non-metals. As closely akin as all the noble gases are to each other, they should surely be considered a separate grouping.
Metals
Past far the largest category of elements on the Periodic Nautical chart is the metal elements. Metals share a set of properties that are non as universal to them equally the inert gases. Metal elements usually have the following properties: They have 1, two, or three electrons on the outside electron trounce.
The outside electrons brand information technology more likely that the metal will lose electrons, making positive ions. The ions of metals are commonly plus 1, plus two, or plus three in charge. Metals tend to lose electrons to become stable. They will adhere to other elements with ionic bonds almost exclusively.
When metal atoms are together in a grouping, at that place is a swarm of semi-loose electrons around the atoms. These electrons movement virtually freely among the metal atoms making what is called an electron gas. The electron gas accounts for the shininess of metals. When at that place is a smoothen surface on the metal it volition reverberate electromagnetic waves (to include visible light) in an organized manner. The shininess is as well called metal luster. The aforementioned electron gas accounts for the cohesive tendencies of metals. Cohesive means the material clings to itself. This property tin exist easily seen with mercury. Mercury atoms cling to other mercury atoms or other metal atoms with an incredible tenacity.
This same cohesion of metals occurs in the solid state. Silver is very malleable. That means that if you hit it, the textile would more likely change shape than shatter. At i time US half dollar coins were made of ninety pct silver. It is illegal to deface money, simply school children would take a spoon and beat out the sides of the argent half dollars until the edges curled inward. When the center became the correct size, it was taken out to make a silver band beaten to fit your finger. Wire is fabricated by pulling metals through a die. The metal coheres to itself so much that it volition reshape itself to the shape of the die every bit it passes through the pigsty in the dice. This property of being able to be pulled through a die to make wire is called ductility (from Latin ducere, to pull or to lead). The presence of the electron gas makes metals skilful conductors of electricity. Once more due to the cohesive property, metals have high melting and boiling points. Almost all metals are solids at room temperature. Metals are usually practiced conductors of heat. Active metals react with acids. Some very active metals will react with water. Metal elements tend to be denser than non-metals.
Non-Metals
The properties of non-metals are non as universal to them as the metals; there is a great bargain of variation among this group. Non-metals take the post-obit properties:
- Non-metals commonly take four, v, half dozen, or seven electrons in the outer shell. When they bring together with other elements non-metals can either share electrons in a covalent bond or gain electrons to become a negative ion and make an ionic bail.
- When non-metallic elements bring together by covalent bonds, it is unremarkably to other non-metals. Non-metals tin attach together with covalent bonds to make a group of (usually non-metal) elements with a mutual accuse called a radical or polyatomic ion.
- Elemental non-metals often accept a dull advent. They are more probable to be brittle, or shatter when struck. Although non a constant rule, not- metals tend to have lower melting and humid points than metals and the solids tend to exist less dense.
- Non-metals are not as cohesive as metals and certainly not ductile. Non-metals are not usually good conductors of heat or electricity. Many non-metals form diatomic or polyatomic molecules with other atoms of the same element.
- Many non-metals have more than than one grade of the free chemical element, called allotropes, that appear in different conditions. (The give-and-take free hither ways that the element is unattached to other types of cantlet, not that information technology has a monetary value of zip.)
Semi-Metals
We take pretended that there is a sharp dividing line between the metals and non-metals. This is not the case. The staircase-shaped line betwixt metals and not-metals has several elements on or near it that have properties somewhere between the 2 categories. By having three electrons in the exterior shell, boron should be a metallic element. It is not. Boron is more likely to form covalent bonds like a non-metal than donate electrons like aluminum, the next element down the chart in the same group.
Aluminum is definitely a metal in most of its traits, but information technology has its own idiosyncrasy. Aluminum is amphoteric; information technology reacts with both acids and bases. Silicon, germanium, arsenic, antimony, and tellurium are on the line between metals and non-metals and exhibit some of the qualities of both. These elements do non actually incorporate a clear-cut category, merely, due to the mix of backdrop they show, they are oft lumped into a classification called semi-metals.
Many of the elements on the line are semiconductors of electricity, meaning that they have the ability to conduct electricity somewhere between near none and total conduction. This property is useful in the electronics manufacture.
Hydrogen
We have failed to include hydrogen in any of the categories, for good reasons. Hydrogen simply does non lucifer annihilation else. More than than ninety-nine-point-nine percent of hydrogen is just one proton and one electron.
A very small proportion (1 cantlet in several thousand) of hydrogen is deuterium, ane proton, ane neutron, and one electron. An even smaller portion (one hundred atoms per million billion) of hydrogen is tritium, one proton, 2 neutrons, and one electron. When a hydrogen atom gains an electron, it becomes a negative ion. The negative hydrogen ion, called hydride ion, can be attached to metals, but information technology is not seen in nature because information technology is not stable in h2o. The positive hydrogen ion is what is responsible for acids. There actually is no such thing as a (positive) hydrogen ion. Having only a proton and an electron, hydrogen becomes only a proton if information technology loses its electron. Loose protons attach themselves to a h2o molecule to brand H3O+ ion, a hydronium ion. This hydronium is the real chemical that produces the properties of acids. Elemental hydrogen is a diatomic gas. Except for having a valence of +1, hydrogen has few other similarities with the Group 1 elements. Hydrogen makes covalent bonds between other hydrogen atoms or other non-metals.
Groups or Families of the Periodic Nautical chart
Group I (1) elements, lithium, sodium, potassium, rubidium, cesium, and francium, are also called the alkali metallic elements. They are all very soft metals that are not found costless in nature because they react with water. In the element form they must exist stored nether kerosene to keep them from reacting with the humidity in the air. They all take a valence of plus one because they have 1 and just one electron in the outside shell. All of the alkali metals show a distinctive color when their compounds are put into a flame. Spectroscopy (dividing up the spectrum so you can see the individual frequencies) of the colored low-cal from the flame examination shows strong emission lines from the elements. The lightest of them are the least reactive. Activity increases as the element is further down the Periodic Chart. Lithium reacts leisurely with water. Cesium reacts very violently. Very few of the salts of Group 1 elements are not soluble in water. The lightest of the alkali metals are very common in the globe'southward crust. Francium is both rare and radioactive.
Group II (2) elements, beryllium, magnesium, calcium, strontium, barium, and radium, all accept two electrons in the outside band, and so have a valence of 2. Besides chosen the alkaline globe metals, Group 2 elements in the complimentary form are slightly soft metals. Magnesium and calcium are common in the crust of the earth.
Group iii elements, boron, aluminum, gallium, indium, and thallium, are a mixed group. Boron has mostly not- metal properties . Boron will bond covalently by preference. The residue of the group are metals. Aluminum is the but one common in the earth's crust. Group iii elements take three electrons in the outer crush, but the larger iii elements take valences of both 1 and three.
Group 4 elements, carbon, silicon, germanium, tin, and lead, are not a coherent grouping either. Carbon and silicon bond nearly exclusively with iv covalent bonds. They both are common in the globe's crust. Germanium is a rare semi-metal. Tin can and atomic number 82 are definitely metals, fifty-fifty though they have four electrons in the outside vanquish. Tin and pb accept some differences in their properties from metallic elements that suggest the short distance from the line between metals and non-metals (semi-metal weirdness). They both have more than one valence and are both somewhat common in the crust of the earth.
Group 5 is also carve up betwixt metals and non-metals. Nitrogen and phosphorus are very definitely not-metals. The chemical element nitrogen equally a diatomic molecule forms about eighty percent of the atmosphere. In the rare instances that nitrogen and phosphorus form ions, they grade triple negative ions. Nitride (Due north-3) and phosphide (P-3) ions are unstable in water, and and then are not constitute in nature. All of the Group 5 elements have 5 electrons in the outer shell. For the smaller elements it is easier to complete the trounce to become stable, and then they are non-metals and are more probable to form covalent bonds than ionic bonds. The larger elements in the group, antimony and bismuth, tend to be metals because it is easier for them to donate the 5 electrons than to attract three more. Arsenic, antimony and bismuth have valences of +3 or +5. Arsenic is very much a semi-metal, but all three of them show some semi-metallic weirdness, such as brittleness every bit a free element.
Group VII (vi or 16) elements, oxygen, sulfur, selenium, and tellurium, have half-dozen electrons in the outside crush. We are not concerned with polonium as a Group 6 chemical element. It is too rare, too radioactive, and too dangerous for us to even consider in a bones grade.
Tellurium is the only chemical element in Group vi that is a semi-metallic. In that location are positive and negative ions of tellurium. Oxygen, sulfur, and selenium are true not-metals. They have a valence of negative two as an ion, simply they also bail covalently. Oxygen gas makes covalent double-bonded diatomic gas molecules that are near 20 percent of the earth's atmosphere.. Oxygen and sulfur are mutual elements. Selenium has a belongings that may exist from semi-metallic weirdness; it conducts electricity much better when light is shining on it. Selenium is used in photocells for this property.
On some charts you will encounter hydrogen above fluorine in Group Vii (7 or 17). Hydrogen does non belong at that place any more than information technology belongs above Group 1. Fluorine, chlorine, bromine, and iodine make up Group 7, the halogens. We tin can forget near astatine. It is too rare and radioactive to warrant any consideration hither. Halogens have a valence of negative i when they make ions because they accept seven electrons in the outer shell. They are all diatomic gases equally free elements near room temperature. They are choking poisonous gases. Fluorine and chlorine are yellow-green, bromine is red, and iodine is purple as a gas. All tin be found fastened to organic molecules. Chlorine is common in the world's crust, much of information technology as the negative ion of common salt, NaCl, in the oceans. Fluorine is the most active of them, and the activity decreases as the size of the halogen increases.
The inert gases or noble gases all accept a complete outside shell of electrons. Helium is the only 1 that has only an "s" subshell filled, having only two electrons in the outer and but shell. All the others, neon, argon, krypton, xenon, and radon, have viii electrons in the outer beat. Since the electron configuration is nigh stable in this shape, the inert gases do not form natural compounds with other elements. The group is variously numbered as Group VIIIA, 8, 8A, 0, or 18. 'Group zero' seems to fit them nicely since information technology is piece of cake to recollect of them every bit having a naught valence, that is no likely charge.
The Transition Elements brand up a group between what Chemtutor has labeled Group two and Group 3. Transition elements are all metals. Very few of the transition elements have any non-metal properties. Inside the transition elements many charts subdivide the elements into groups, merely other than three horizontal groups, it is difficult to brand meaningful distinctions amid them. The horizontal groups are: fe, cobalt, and nickel; ruthenium, rhodium, and palladium; and osmium, iridium, and platinum. Iron is idea to be plentiful equally a molten mass in the center of the globe.
Lanthanides and actinides are called the Inner Transition Elements. Lanthanides, elements 57 through 70, are also chosen the rare globe elements. They are all metal elements very similar to each other, but may exist divided into a cerium and a yttrium group. They are oft found in the same ores with other elements of the group. None are establish in whatever great quantity in the earth'south chaff. Of the Actinides, elements 89 through 102, only the first iii are naturally occurring, the rest existence manufactured elements. Of the three naturally occurring ones, only uranium is likely to be referred to in any way in a bones chemistry form. Elements 103 through 109 take been manufactured, and they have been named past the IUPAC (International Union of Pure and Practical Chemical science), but they are non of much importance to this course.
The Familiar Periodic Tabular array with Atomic Numbers, Symbols, and Masses
| Avogadro's Number | N = 6.022 E23 |
| Gas Abiding | R = 0.08206 liter atm/mole – Yard = 8.3145 J/mole – One thousand |
| Planck'southward Constant | h = six.626 E-34 J – sec |
| Velocity of Light | c = 2.998 E8 1000/sec |
| Faraday Constant | F = 96,485 coul/mole of e'southward |
| Electric Current | Amperes = coulombs/seconds |
| Electrical Free energy | Joules = volts x coulombs |
| piece of work value of heat | 1 calorie = 4.184 J |
| Specific Estrus Chapters of Liquid Water | c H2o = iv.18 J/g – C° = one.00 cal/ k – C° |
| | | ||||||||||||||||
| | | | | 5-A | Six-A | | | ||||||||||
| | | | | | | | | | |||||||||
| | | | | | | | | | | | | | | | | ||
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | |
| | | | | | | | | | (269) | | | | | | |||
| | | 140.908 | | | | 151.964 | | | | | | | | |
| | | | | | | | | | | | | | |
Source: https://www.wyzant.com/resources/lessons/science/chemistry/periodic-table/
0 Response to "Which Combination of Elements Represents Part of a Group, or Family, of the Periodic Table"
Post a Comment